|
Forecast
Period
|
2026-2030
|
|
Market
Size (2024)
|
USD
1625.85 Million
|
|
Market
Size (2030)
|
USD
2240.63 Million
|
|
CAGR
(2025-2030)
|
5.45%
|
|
Fastest
Growing Segment
|
Active
Pharmaceutical Ingredient (API)
|
|
Largest
Market
|
Northern
& Central
|
Market Overview
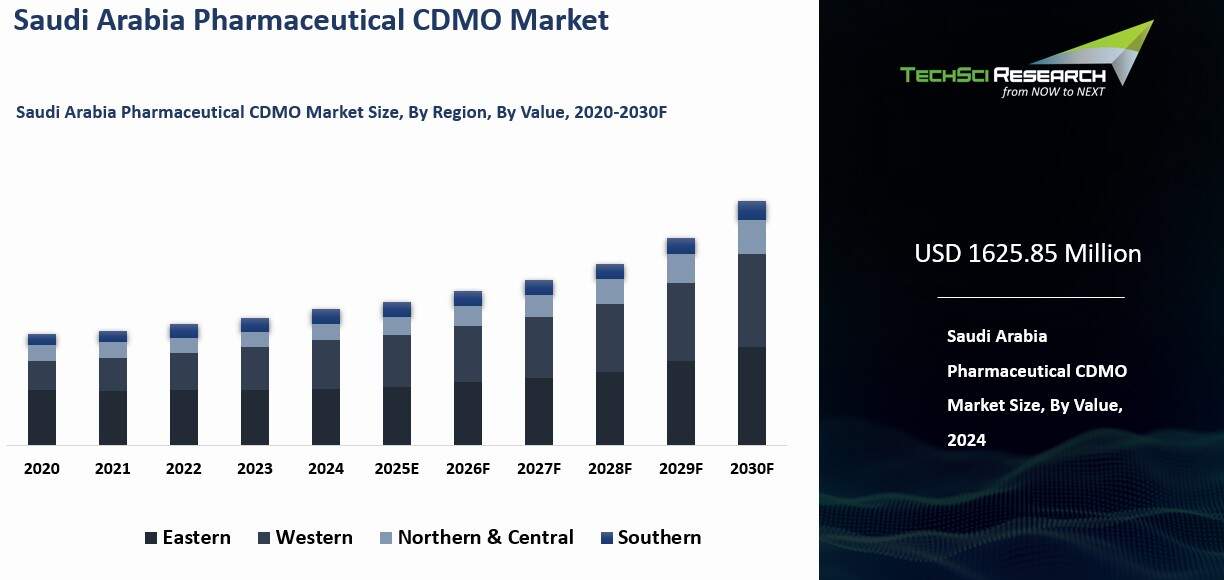
Saudi Arabia Pharmaceutical CDMO Market was valued at USD 1625.85 Million in 2024 and is expected to reach USD 2240.63 Million by 2030 with a CAGR of 5.45% during the forecast period.
Vitamin supplements are designed to enhance the regular diet by providing individuals with the necessary daily nutritional value. Vitamins play crucial roles in the development and proper functioning of the body, acting as hormones, coenzymes, and antioxidants. Various factors such as shifting dietary preferences, busy lifestyles, rising employment rates, and increased awareness of the health benefits associated with vitamin supplements are expected to positively influence the market growth.
Due to hectic schedules, many individuals struggle to maintain a balanced diet, resulting in nutrient deficiencies. Consequently, there has been a significant rise in the consumption of vitamin supplements to fulfill daily nutrient and vitamin requirements, promoting overall health and vitality.
The increasing healthcare expenditure worldwide is also anticipated to drive the demand for vitamin supplements. Additionally, the growing elderly population in both developed and developing economies presents lucrative opportunities for market players in the forecast period. The senior population, in particular, relies on vitamin supplements to meet their dietary needs, promote bone health, and support overall well-being.
Key Report Takeaway
- Active Pharmaceutical Ingredient segment led in 2024, supported by rising generic drug demand, affordability priorities, and high prescription volumes across the Kingdom.
- Vision 2030 localization strategy strengthened API manufacturing, reducing import reliance, improving supply security, and enabling export readiness through incentives and funding support.
- Operational efficiency favored API production, as lower capital intensity, scalable capacity expansion, and economies of scale enhanced pricing competitiveness for Saudi CDMOs.
- Northern and Central regions dominated, led by Riyadh with strong logistics links to Dammam and Jeddah ports, modern industrial zones, and advanced pharmaceutical manufacturing clusters.
Download Free Sample Report
Key Market Drivers
Rising Burden of Chronic Diseases and Domestic Demand
Saudi Arabia is seeing stronger domestic pharmaceutical demand as chronic conditions become more common and patients access care more consistently through an expanding healthcare network. Drug makers face sustained pressure to keep essential medicines and specialty therapies available at high volumes, especially as payers and providers push earlier intervention and tighter follow-up for long-term diseases.
For instance, the International Diabetes Federation estimates Saudi Arabia had about 5,344,600 adults ages 20 to 79 living with diabetes in 2024 and about 2,330,200 adults with undiagnosed diabetes, which supports steady demand for screening, treatment initiation, and chronic medication supply. Partnering with CDMOs helps developers avoid capacity constraints, stabilize supply planning, and deliver medicines reliably as localized demand expands.
Strategic Shift Toward Biologics and Oncology Treatments
The Saudi CDMO landscape is shifting beyond conventional small-molecule manufacturing toward biologics, biosimilars, and advanced therapies that require specialized infrastructure, validated sterile processes, and stricter quality systems. Oncology and immunology pipelines also raise complexity because they depend on high-containment practices, tight cold-chain controls, and rigorous batch release oversight aligned with international GMP expectations.
For instance, the Saudi Food and Drug Authority reported clinical trial applications for advanced therapies and biotechnology products increased 83% in 2025, about 2,700 patients benefited from participation in these trials, and its Reliance Pathway for Clinical Trials reduced approval time by 74% versus the standard track, signaling faster-moving innovation that increases demand for capable outsourcing partners. As sponsors prioritize speed and compliance, technologically advanced CDMOs become essential for scaling complex production while maintaining quality consistency.
Health System Modernization and Infrastructure Expansion
Saudi Arabia’s health system modernization is expanding the footprint of care delivery and strengthening demand for dependable supply across routine therapies, specialized regimens, and clinical research materials. Hospital expansion, digitized records, and national screening and prevention campaigns increase prescription throughput and intensify the need for uninterrupted availability of diverse dosage forms, including injectables and other specialized products. For instance, the Ministry of Health said Seha Virtual Hospital delivered more than 16 million virtual appointments and consultations in 2025 and managed more than 220,000 clinical cases, showing how digital care models can increase continuity of care and medication demand across wider catchments.
CDMOs are positioned to provide the agility and scale needed to serve newly expanding facilities and more integrated care pathways with consistent, compliant production.
Key Market Challenges
Regulatory and Compliance Barriers
Regulatory complexity remains a constraint for Saudi Arabia’s pharmaceutical CDMO sector, despite recent reforms. The Saudi Food and Drug Authority has introduced measures to improve transparency and shorten review timelines, yet approval pathways for new formulations and advanced therapies can still be lengthy. Delays affect time to market and reduce the speed at which innovative products reach patients.
CDMOs must comply with domestic regulations while also meeting standards set by authorities in the United States, Europe, and Asia. Dual compliance increases operational costs, documentation requirements, and audit exposure. Any deviation from global benchmarks may lead to shipment holds, restricted market access, or financial penalties. This environment limits scalability and affects competitiveness in international supply chains.
Shortage of Skilled Labor and Expertise
Pharmaceutical manufacturing depends on expertise in biotechnology, regulatory affairs, quality assurance, and process engineering. Saudi Arabia continues to face a shortage of highly specialized professionals in these fields. While the domestic talent pool is expanding, advanced technical capabilities required for biologics and complex therapies remain limited.
Workforce gaps can slow technology adoption, automation integration, and large scale production expansion. Reliance on expatriate professionals raises labor costs and adds retention challenges. Although government backed education and training initiatives are underway, the pace of skill development may not fully match sector growth, creating ongoing operational pressure for CDMOs seeking global competitiveness..
Key Market Trends
Integration of Advanced Manufacturing Technologies
Saudi Arabia’s pharmaceutical manufacturing future is increasingly shaped by automation, artificial intelligence, and data analytics. CDMOs are adopting automated production lines to improve efficiency, reduce manual errors, and maintain consistent product quality. Automation supports faster batch cycles, better scalability, and stronger cost control, which are essential for both domestic supply and export readiness.
AI and machine learning enhance process optimization, predictive maintenance, and real time quality monitoring. By analyzing large production datasets, manufacturers can identify deviations early, forecast equipment failures, and improve yield performance. These tools are especially important in complex manufacturing environments, including biologics, where formulation accuracy and sterile conditions are critical. Integration of digital systems also strengthens traceability and compliance, aligning with standards set by the Saudi Food and Drug Authority.
Shift Towards Biopharmaceuticals and Specialty Drugs
The pharmaceutical landscape is moving toward biologics, biosimilars, gene therapies, and targeted treatments, and Saudi Arabia is aligning with this shift. Rising prevalence of chronic and rare diseases is increasing demand for advanced therapies that require specialized manufacturing capabilities. CDMOs are expanding infrastructure for aseptic processing, cold chain logistics, and bioreactor based cell cultivation to support biologic production.
Government priorities under Vision 2030 emphasize reducing import reliance and strengthening local production of high value medicines. This strategy positions Saudi Arabia as a developing regional hub for specialty drug manufacturing. As global pharmaceutical firms expand outsourcing of complex products, Saudi CDMOs are gaining opportunities to serve as cost efficient and compliant partners, reinforcing long term sector growth.
.jpg)
Download Free Sample Report
Segmental Insights
Product Insights
In 2024, the Active Pharmaceutical Ingredient segment held the largest share of Saudi Arabia’s pharmaceutical CDMO sector. Growth is strongly linked to rising demand for generic medicines across the Kingdom and the wider Middle East. As healthcare systems focus on cost control and broader access to essential treatments, generics remain a priority due to affordability and high prescription volumes.
Saudi CDMOs engaged in API manufacturing supply key inputs for a wide range of generic drugs, strengthening domestic availability and reducing reliance on imported raw materials. Localization efforts under Vision 2030 support investment in large scale API facilities through tax incentives, subsidies, and funding support. This policy direction enhances supply chain security and improves export readiness within regional markets.
API production also offers operational advantages compared with full drug formulation. It generally involves lower capital intensity and fewer downstream regulatory requirements, allowing faster capacity expansion. Economies of scale improve pricing competitiveness, enabling Saudi CDMOs to offer cost efficient, high quality active ingredients. Rising generic consumption, supportive government policy, and scalable production economics are expected to sustain API segment leadership in the coming years.
Regional Insights
In 2024, the Northern and Central regions held the largest share of Saudi Arabia’s pharmaceutical CDMO sector in both value and volume terms. Riyadh serves as the national business and logistics center, supported by strong transport corridors and industrial infrastructure. Its central position allows efficient distribution across domestic markets and streamlined coordination with international partners.
Proximity to major ports such as Dammam and Jeddah strengthens supply chain efficiency by facilitating raw material and API imports while enabling exports of finished formulations. This connectivity supports CDMOs targeting the Middle East, North Africa, and parts of Asia, reinforcing the region’s logistical advantage within regional trade routes.
Modern industrial zones in Riyadh and surrounding areas host advanced pharmaceutical manufacturing facilities aligned with international standards. High tech plants support production of small molecule drugs, biologics, and specialized therapies. Dedicated pharmaceutical clusters improve regulatory coordination, operational scalability, and compliance efficiency, sustaining the Northern and Central regions’ leadership in the national CDMO landscape.
Recent Developments
- In October 2025, WuXi AppTec announced it signed two strategic MoUs with NEOM and Saudi Arabia’s Ministry of Health to explore localizing pharmaceutical research, development, and manufacturing in the Kingdom, including laying groundwork for CRDMO facilities at Oxagon and initiatives around localizing the pharma value chain and workforce development.
- In April 2025, Polaris Pharmaceuticals signed an exclusive licensing and supply agreement with Tabuk Pharmaceuticals to introduce ADI-PEG20 across Saudi Arabia and the GCC. Under this structure, Polaris oversees development and manufacturing, while Tabuk manages marketing authorization and commercialization reflecting a CDMO-linked supply and tech-transfer model.
- In March 2024, the Public Investment Fund (PIF) launched Lifera, a national biopharma CDMO platform designed to expand vaccine, biologics, and broader biomanufacturing capabilities, establishing sovereign-backed contract development and manufacturing within Saudi Arabia.
- In August 2024, SPIMACO entered a regional partnership with Altos Biologics to locally manufacture and commercialize ALT-L9 (aflibercept biosimilar), advancing technology transfer and regional production of specialty biologics in the Kingdom.
- In
December 2024, Bio-Thera Solutions Inc, a biopharmaceutical company focused on
developing innovative therapies and biosimilars, has entered into a strategic
partnership with Tabuk Pharmaceutical Manufacturing Company, a fully owned
subsidiary of Astra Industrial Group and a leading pharmaceutical player in the
Middle East and North Africa (MENA) region. Under the terms of the agreement,
Tabuk has secured exclusive rights to manufacture, distribute, and market
BAT2206, Bio-Thera’s ustekinumab biosimilar, in Saudi Arabia. This partnership
aims to expand the availability of advanced biologic treatments in the region
while enhancing Tabuk's portfolio with a key biosimilar offering.
Key Market Players
|
By
Product
|
By
Application
|
By
Workflow
|
By
Region
|
- API
- Synthetic
- Solid
- Liquid
- Biotech
- Drug Product
- Oral Solid Dose
- Semi-solid Dose
- Liquid Dose
- Others
|
- Oncology
- Small Molecule
- Biologics
- Infectious Diseases
- Neurological Disorders
- Cardiovascular Diseases
- Metabolic Disorders
- Autoimmune Diseases
- Respiratory Diseases
- Ophthalmology
- Gastrointestinal Disorders
- Hormonal Disorders
- Hematological Disorders
- Others
|
|
- Eastern
- Western
- Northern & Central
- Southern
|
Report Scope:
In this report, the Saudi Arabia Pharmaceutical
CDMO Market has been segmented into the following categories, in addition to
the industry trends which have also been detailed below:
- Saudi Arabia Pharmaceutical CDMO Market, By Product:
o API
o Synthetic
o Solid
o Liquid
o Biotech
o Drug Product
o Oral Solid Dose
o Semi-solid Dose
o Liquid Dose
o Others
- Saudi Arabia Pharmaceutical CDMO Market, By Application:
o Oncology
o Small Molecule
o Biologics
o Infectious Diseases
o Neurological Disorders
o Cardiovascular Diseases
o Metabolic Disorders
o Autoimmune Diseases
o Respiratory Diseases
o Ophthalmology
o Gastrointestinal Disorders
o Hormonal Disorders
o Hematological Disorders
o Others
- Saudi Arabia Pharmaceutical CDMO Market, By Workflow:
o Clinical
o Commercial
- Saudi Arabia Pharmaceutical CDMO Market, By
Region:
o Eastern
o Western
o Northern & Central
o Southern
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Saudi
Arabia Pharmaceutical CDMO Market.
Available Customizations:
Saudi Arabia
Pharmaceutical CDMO market report with the given market data, Tech Sci
Research offers customizations according to a company's specific needs. The
following customization options are available for the report:
Company Information
- Detailed analysis and profiling of additional
market players (up to five).
Saudi Arabia Pharmaceutical CDMO Market is an
upcoming report to be released soon. If you wish an early delivery of this
report or want to confirm the date of release, please contact us at [email protected]